- Blog
- Download lagu taylor swift 22 mp3
- Hp 2025 printer fuse parts names
- Cat ninja wanted book 2
- Cacl2 molar mass
- Strategy guide for black ops zombies kino der toten
- Nicecast shoutcast
- Minecraft pocket edition download for kindle fire
- Bank writedown
- Uninstall remove cyberpower powerpanel personal
- Wowmatrix windows 7
- Macminer setup
- The insidious 3 full movie download
- Outlook for mac search email attachments
- Pc translator and dictionary
- Pure color 211 aloof lipstick
- Air freshener camera recorder
- Emcee trucking

#Cacl2 molar mass how to#
The solution can be stored at room temperature or in a cold room. Explanation of how to find the molar mass of CaCl2: Calcium chloride.A few things to consider when finding the molar mass for CaCl2:- make sure you have the. It is highly soluble in water and it is deliquescent. Calcium chloride is an ionic compound of calcium and chlorine. Line 2 Required work shown: Calculate the molar mass of Calcium chloride dihydrate. Calcium dichloride is a calcium salt, an inorganic chloride and an inorganic calcium salt. Sterilize the solution by autoclaving (20 minutes at 15 lb/sq.in. We use the molar ratio of reactant in a balanced chemical reaction to.

Step 3: Transfer the medium to an autoclavable bottle.
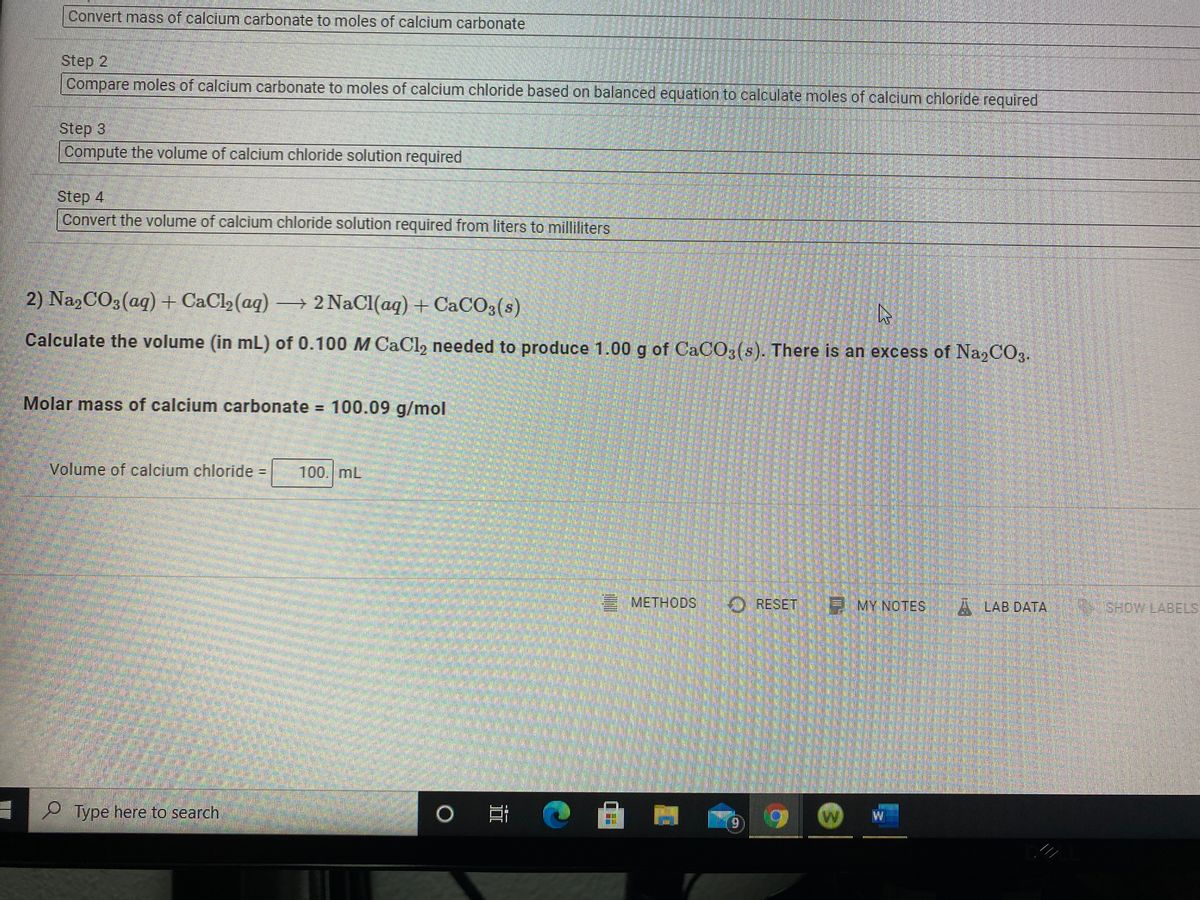
Step 2: Adjust the volume to 1000 ml with deionized water. ◊ Make sure to dissolve any residual powder sticking to the glass. Question: What mass of calcium chloride, CaCl2 (molar mass 111.0 g/mol) is needed to prepare 2.850 L of a 1. In most cases, solution volume increases when a large amount of solute dissolves in the solvent. It is always a good practice to dissolve all media ingredients first and then adjust the volume to the desired volume with the solvent. ◊ Do not dissolve in 1000 ml of deionized / Milli-Q water. S.I Unit of Molar mass is gram per mole and it is.2 answers Top answer: Molecular mass of texCaCl2/tex is 111 g Explanation:Molecular mass is defined as the. Composition: Calcium Chloride 100 Grade: Anhydrous, Purified Boiling Point: 1670C CAS Number: 1 Density: 2.15 Chemical Formula: CaCl2 Molecular. CaCl.1 answer Top answer: Calcium chloride formula, also known as Calcium dichloride formula or Calcium chloride anhydrous formula, is given in this article. Explanation: Molecular mass is defined as the mass in grams of 1 mole of a substance.
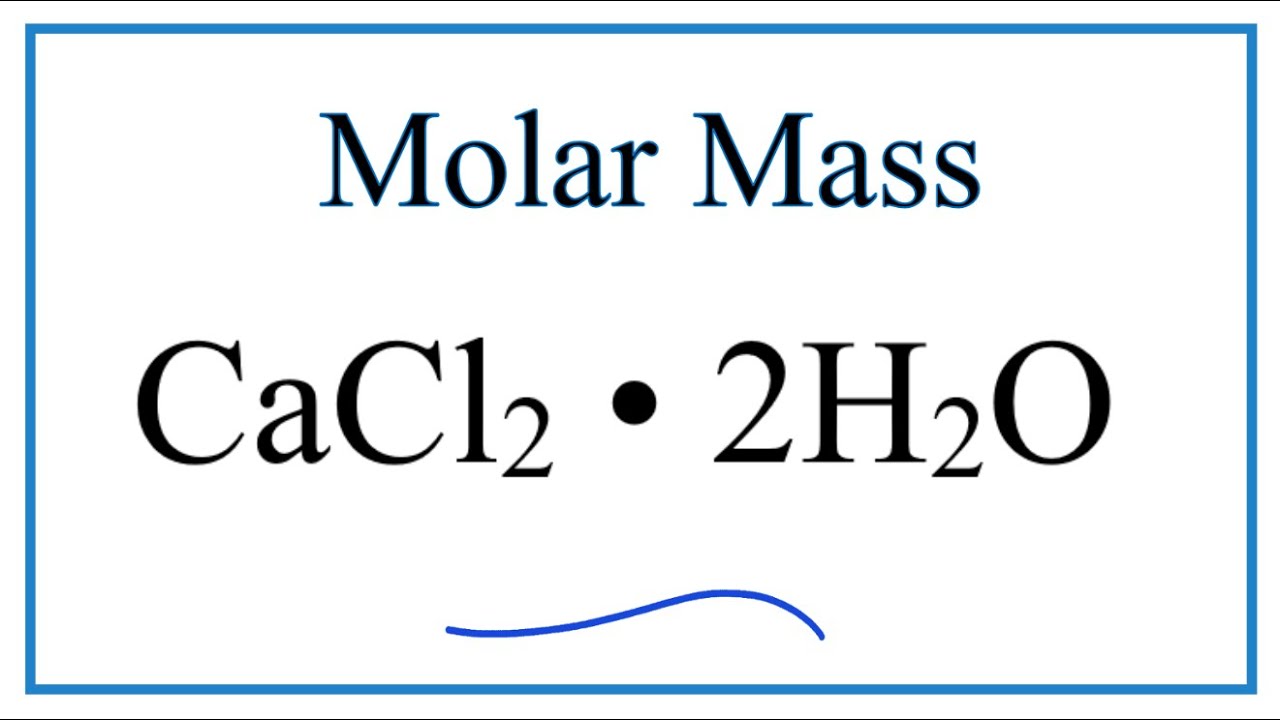
Therefore, a sample of calcium chloride contains twice as many chloride ions as calcium ions. The magnetic stirrer makes the dissolving process easy and convenient. There exist one calcium atom and two chlorine atoms in the structure of calcium chloride. Calcium Chloride is an ionic compound with the chemical formula CaCl2. Mixing using a Magnetic stirrer is optional. Accurate experimental molecular mass values of bovine -trypsin were first.
#Cacl2 molar mass manual#
◊ One can use manual shaking using a glass pipette to mix all ingredients. Step 1: To prepare 1000 ml of 1 M calcium chloride (CaCl2) solution, weigh out 147.02 g of CaCl2.2H2O (Molecular weight: 147.02) and dissolve in 800 ml deionized / Milli-Q water using magnetic stirrer.
